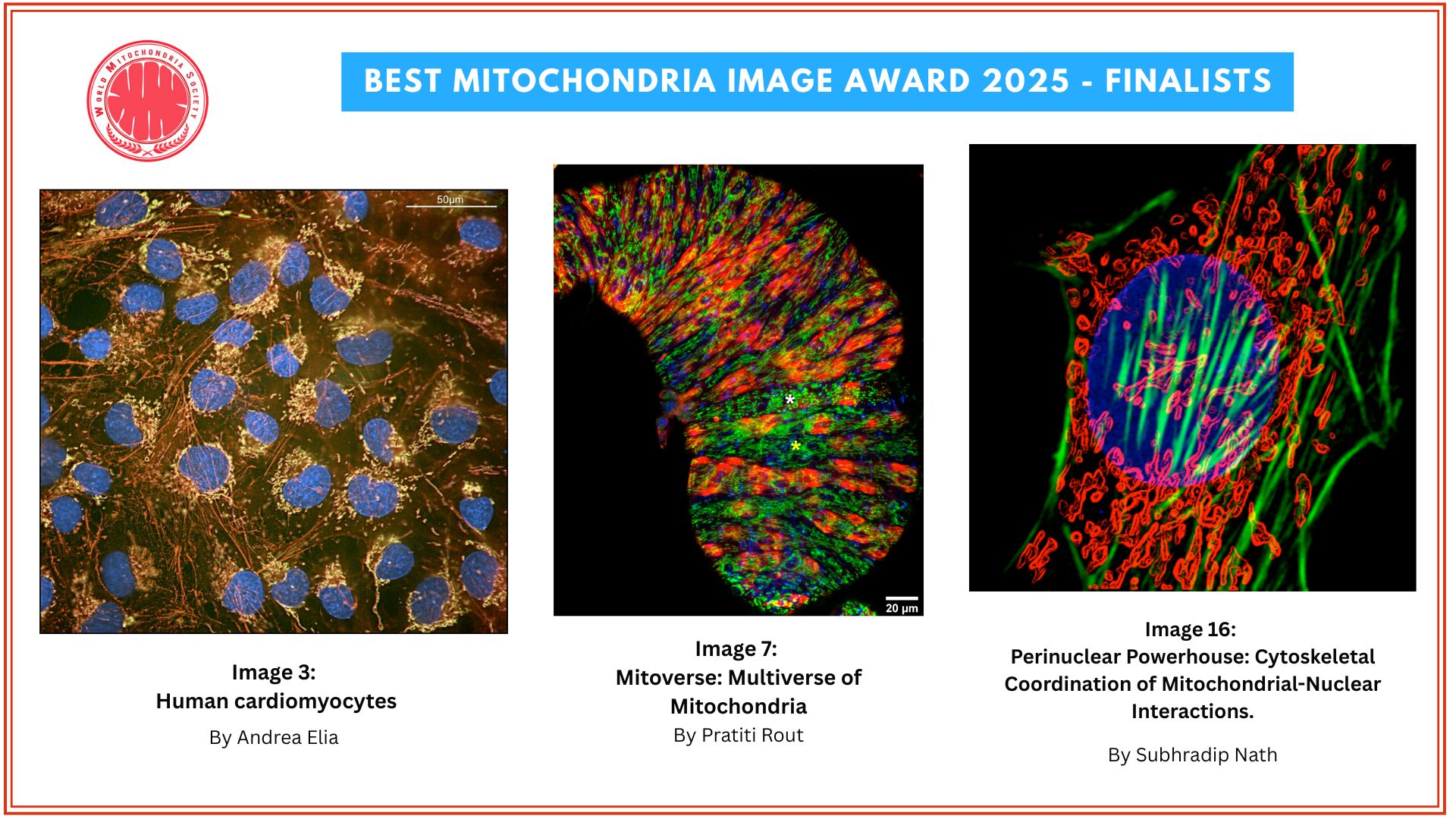
Red Blood Cells Lose Their Mitochondria: But Not Before Building the Machinery of Oxygen Transport
- Details
- Published on 24 April 2026
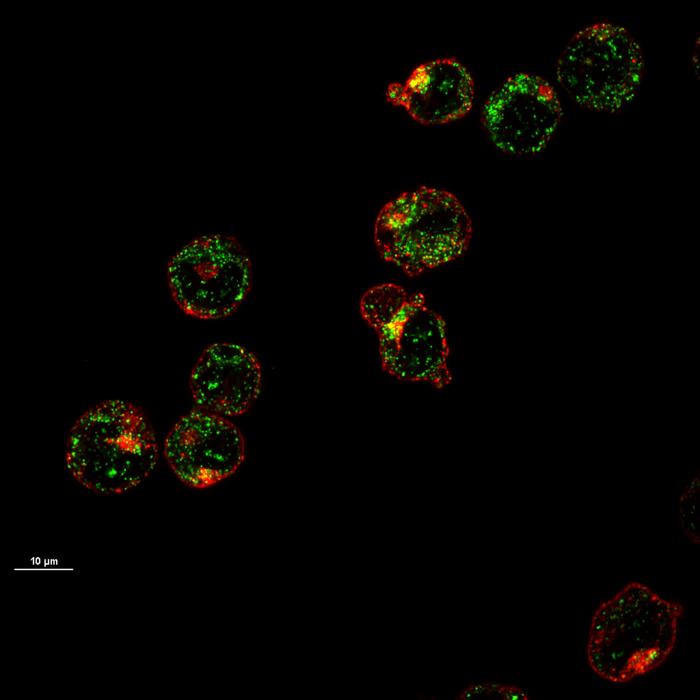
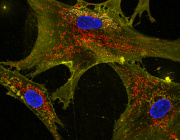
- Maturing precursor red blood cells display Heme Responsive Gene 1(HRG1) transporters (in green). These protein complexes are expressed on cell membranes and shuttle the iron-containing molecule heme from outside to the interior of the cell.
Red Blood Cells Lose Their Mitochondria: But Not Before Building the Machinery of Oxygen Transport
A new study reveals how developing red blood cells import heme to complete oxygen-carrying capacity under stress.
A new study from the University of Maryland School of Medicine led by Iqbal Hamza, published in Science, reveals a surprising mechanism that helps young red blood cells produce hemoglobin when the body is under stress.
Red blood cells carry oxygen through the body using hemoglobin, the red protein that captures oxygen in the lungs and delivers it to tissues. To build hemoglobin, developing red blood cells need heme, an iron-containing molecule that allows oxygen to bind.
Mature red blood cells do not have mitochondria. They lose their mitochondria during development to become highly specialized oxygen carriers. But before they mature, young red blood cells still contain mitochondria, and these mitochondria help produce heme.
This creates an important biological question: How can red blood cells continue building enough hemoglobin while they are losing the mitochondria normally involved in heme production?
The new study identifies a backup pathway. Researchers showed that immature red blood cells, called erythroblasts, can import heme from surrounding cells through a transporter called HRG1. This external heme supply becomes especially important when the body must rapidly produce new red blood cells, such as after blood loss, during anemia, or under low oxygen conditions.
In mouse models, when HRG1 was removed, developing red blood cells could not accumulate enough hemoglobin, failed to mature properly, and the animals became anemic under stress. The researchers also found that modifying HRG1 activity improved red blood cell production in a model of β-thalassemia, an inherited disorder affecting hemoglobin formation.
For mitochondrial biology, this discovery is important because it highlights a transition phase in red blood cell development. Mitochondria help young blood cells produce heme, but as these cells mature and remove their mitochondria, they may rely on heme supplied from outside the cell to complete hemoglobin production.
This finding changes the way we understand blood formation. Hemoglobin production is not only an internal process driven by mitochondria. It also depends on communication and material exchange between cells.
The discovery opens new perspectives for anemia, β-thalassemia, sickle cell disease, and other conditions where heme balance, oxidative stress, and red blood cell production are disrupted.
It also reinforces a broader concept central to mitochondrial medicine: mitochondria are not only energy producers. They participate in the construction, adaptation, and resilience of cells during stress.
Take-home message
Young red blood cells use mitochondria to help make heme. Mature red blood cells no longer have mitochondria. This study shows that, during the transition, developing blood cells can import heme from nearby cells through HRG1 to finish building hemoglobin, especially when the body is under stress.
Read the complete news here: www.eurekalert.org
Article: Audrey Belot et al., A cell-nonautonomous heme acquisition pathway enables erythroid hemoglobinization under stress.Science392, 388-394(2026). DOI:10.1126/science.aea0552