Lucid Scientific Joins as Official Supporter of World Mitochondria Society Congress 2026
- Details
- Published on 05 May 2026

Mitochondrial Research Needs a Shift: From Snapshots to Dynamics
The World Mitochondria Society welcomes Lucid Scientific as a supporter of the 2026 World Congress in Berlin, October 21–23.
Mitochondrial science is moving toward functional, real-time biology. Tools that capture dynamic cellular behavior are now essential.
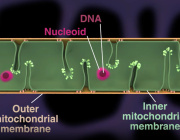
Lucid Scientific’s platform, Resipher, enables continuous, non-invasive measurement of oxygen consumption directly inside standard incubators, with longitudinal profiling over hours to days.
Most metabolic assays still rely on static, endpoint measurements. Mitochondria operate dynamically, adapting continuously to stress, drugs, and signaling inputs.
Continuous oxygen consumption monitoring allows:
- Detection of transient metabolic shifts otherwise missed
- Improved reproducibility across experiments
- Refined analysis of drug response kinetics
- More accurate modeling of mitochondrial adaptation
At the Berlin congress, Lucid Scientific will present how real-time bioenergetics can refine mitochondrial phenotyping and accelerate translational workflows.
Berlin | October 21–23, 2026
For more information about Lucid Scientific and the Resipher system : lucidsci.com.
The Mitochondrial Measurement Challenge: WMS 2026 Workshop
- Details
- Published on 28 April 2026

Before we can target mitochondria, we must first understand how to accurately measure their function, dysfunction, adaptability, and resilience. This high-level workshop will lay the scientific foundation for the congress. This high-level workshop will lay the scientific foundation for the congress. As mitochondrial medicine enters a new era, the key question is no longer only what mitochondria are but how they behave, adapt, fail, and recover.
Led by Naïg Gueguen, the session will explore:
Call for Abstracts & Innovations
- Details
- Published on 15 April 2026

Berlin will host the 17th World Congress of the World Mitochondria Society on October 21–23, 2026. This is more than a call for abstracts. It is a call for ideas, innovations, and translational strategies that move mitochondrial science forward.
From Powerhouse to Prescription: Hazel H. Szeto Joins the WMS As KEYNOTE Speaker
- Details
- Published on 15 April 2026

From Powerhouse to Prescription: Dr. Hazel Szeto Joins the WMS Annual Meeting 2026 as KEYNOTE Speaker. We are proud to announce that Hazel H. Szeto has accepted our invitation to join the WMS Annual Meeting – Targeting Mitochondria 2026, taking place in Berlin on October 21–23, 2026. Dr. Szeto is internationally recognized for pioneering mitochondria-targeted therapeutics and for helping transform mitochondrial science into real medicine. Her work contributed to one of the first FDA-approved mitochondria-targeted therapies a historic milestone for the field.
Welcome to Targeting Mitochondria 2026
- Details
- Published on 01 February 2026

Warm regards,
 Prof. Volkmar Weissig
Prof. Volkmar Weissig
President of the World Mitochondria Society
Midwestern University, USA
Concluding Remarks of WMS 2025
- Details
- Published on 12 January 2026

The 16th World Congress on Targeting Mitochondria marked a defining moment for the mitochondrial community. Over three days in Berlin, the discussions made one thing clear: mitochondria are no longer a niche topic of cell biology, they have become a central pillar of modern medicine. In his concluding remarks, Marvin Edeas, Founder and Chairman of the World Mitochondria Society, emphasized that mitochondrial research has entered a new phase.
WMS 2025 Awards – Celebrating Scientific Excellence in Mitochondrial Medicine
- Details
- Published on 11 January 2026

At Targeting Mitochondria 2025, the World Mitochondria Society honored three outstanding scientific contributions that perfectly reflect the spirit of WMS: bold ideas, rigorous science, and a dynamic vision of mitochondria. These awards celebrate discoveries that connect energy, communication, metabolism, and resilience, reminding us that mitochondria are not static structures, but active players shaping health, disease, and longevity.
Targeting Mitochondria 2025, A Record-Breaking Edition Defining the Next Frontier of Medicine
- Details
- Published on 19 October 2025

The World Mitochondria Society (WMS) proudly announces an unprecedented level of participation at the 17th World Congress on Targeting Mitochondria, uniting more than 237 academic and institutional partners and over 31 industrial and investment organizations.
This year’s edition represents a turning point in mitochondrial science, where fundamental biology meets translational medicine, biotechnology, and AI-driven innovation. From energy metabolism to organelle communication, mitochondria are emerging as strategic integrators of cellular function, systemic health, and longevity.
“Mitochondria are no longer seen only as the cell’s powerhouse,” said Volkmar Weissig and Marvin Edeas, President and Founder of the WMS.
“We are now decoding their language, how they communicate, adapt, and influence other organelles. This knowledge is redefining how we approach diagnostics, therapeutics, and prevention.”
With record-breaking attendance and scientific diversity, Targeting Mitochondria 2025 will feature:
Australia:
· Deakin University
Belgium:
· ORBI team, URBC, University of Namur
Brazil:
· University of Campinas (UNICAMP)
Bulgaria:
· Institute of Biology and Immunology of Reproduction, Bulgarian Academy of Sciences
Canada:
· Lunella Biotech. Inc.,
Chile:
· IMPACT CENTER, Universidad de Los Andes
China:
· Kunming Medical University · Shanghai jiaotong university
Czech Republic:
· Charles University and General University Hospital in Prague · Institute Of Physiology Of The Czech Acad. Sci. · Institute of Physiology, Czech Academy of Sciences
Finland:
· Tampere University · University of Eastern Finland
France:
· CEA MIRCen CNRS UMR 9199 · INSERM INM · INTERNATIONAL SPACE FEDERATION · L'Oreal R&I · Proya Europe · Université paris Cité, INSERM · University Paris-Saclay; INRAE
Germany:
· Albstadt-Sigmaringen University · Charité - Universitätsmedizin Berlin · Deutsches Herzzentrum der Charité · Deutsches Herzzentrum der Charite · Universitätsklinikum Erlangen · Dr. Mühlhausen · HAW Albstadt-Sigmaringen · Universitätsklinikum Essen · University Albstadt-Sigmaringen · University Medical Center Göttingen
Greece:
· EK PA
India:
· Indian Institute of Science Education and Research Tirupati, India · National Institute of Pharmaceutical Education and Research (NIPER) - Ahmedabad
Israel:
· Hebrew University of Jerusalem · Minovia Therapeutics Ltd. · Neurim Pharamceuticals · Sheba Medical Center
Italy:
· Crab Sinergy Srl · IDI IRCCS- Fondazione Maria Luigi Monti · Institute of Cristallography, National Council of Research · IRCCS San Raffaele Scientific Institute · National Research Council (CNR) · Polytechnic University of Marche
· Università degli Studi dell'Insubria · Università degli Studi di Bari · University of Milan · University of Padova · University of Salento · University of Turin · University of Verona
Japan:
· Hokkaido University · Kyowa Kirin · National Cerebral and Cardiovascular Center Research Institute · Tokyo University of Science
Kazakhstan:
· Nazarbayev University
Lithuania:
· Lithuanian University of Health Sciences
Luxembourg:
· University of Luxembourg
Mexico:
· Universidad de Guanajuato
Monaco:
· Biotech Investor
Netherlands:
· Amsterdam UMC · Maastricht University · Radboud University Department of Microbiology · UMC Utrecht
Norway:
· Miphic · University of Oslo
Poland:
· Adam Mickiewicz University in Poznań · Institute of Fundamental Technological Research, Polish Academy of Sciences · Nencki Institute of Experimental Biology PAS · Warsaw University of Life Sciences
· Wroclaw Medical University
Singapore:
· National University of Singapore
Slovakia:
· OVOGENE / IFG
Slovenia:
· University of Ljubljana · University of Ljubljana, Faculty of Pharmacy · University of Maribor
South Korea:
· CHA University · Institute of Endemic Diseases / Seoul National University Medical Research Center · Korea Institute of Science and Technology (KIST) School · Seoul National University College of Medicine
Spain:
· Centro Nacional de Investigaciones Cardiovasculares, Madrid, Spain · Universidad de Zaragoza
· University of Oviedo
Sweden:
· Karolinska Institutet · Stockholm Univerisity
Switzerland:
· ETH Zürich · Novartis Biomedical Research
· University of Basel
Turkey:
· Hacettepe University · Koc University · Kocaeli University, Department of Stem Cell
United Kingdom:
· Altos Labs UK · CellSpex Ltd · King’s College London · Northumbria University, Newcastle University · The Roger Williams Institute of Liver Studies · University of Bath · University of Cambridge
United States:
· Aardvark Therapeutics · ASU School of Molecular Sciences · Ciity of Hope National Medical Center · Colossal Biosciences · Labryo Fertility Center · Michael J Fox Foundation · No affiliation · Social Profit Network · Solid Biosciences · Stanford University · Throne Biotechnologies · University of California Los Angeles · University of California San Diego · University of California San Francisco (UCSF) · University of California, San Diego/ Neurosciences · University of South Dakota · UT Texas MD Anderson Cancer Center · Vertex Pharmaceuticals · Wayne State University · Wayne State University, Detroit · West Virginia University
As mitochondria move from passive subjects of research to dynamic targets for intervention, the 2025 Congress will serve as a global platform to chart the path from observation to transformation, from understanding energy to engineering health.
Discover who is attending and explore the full scientific program.
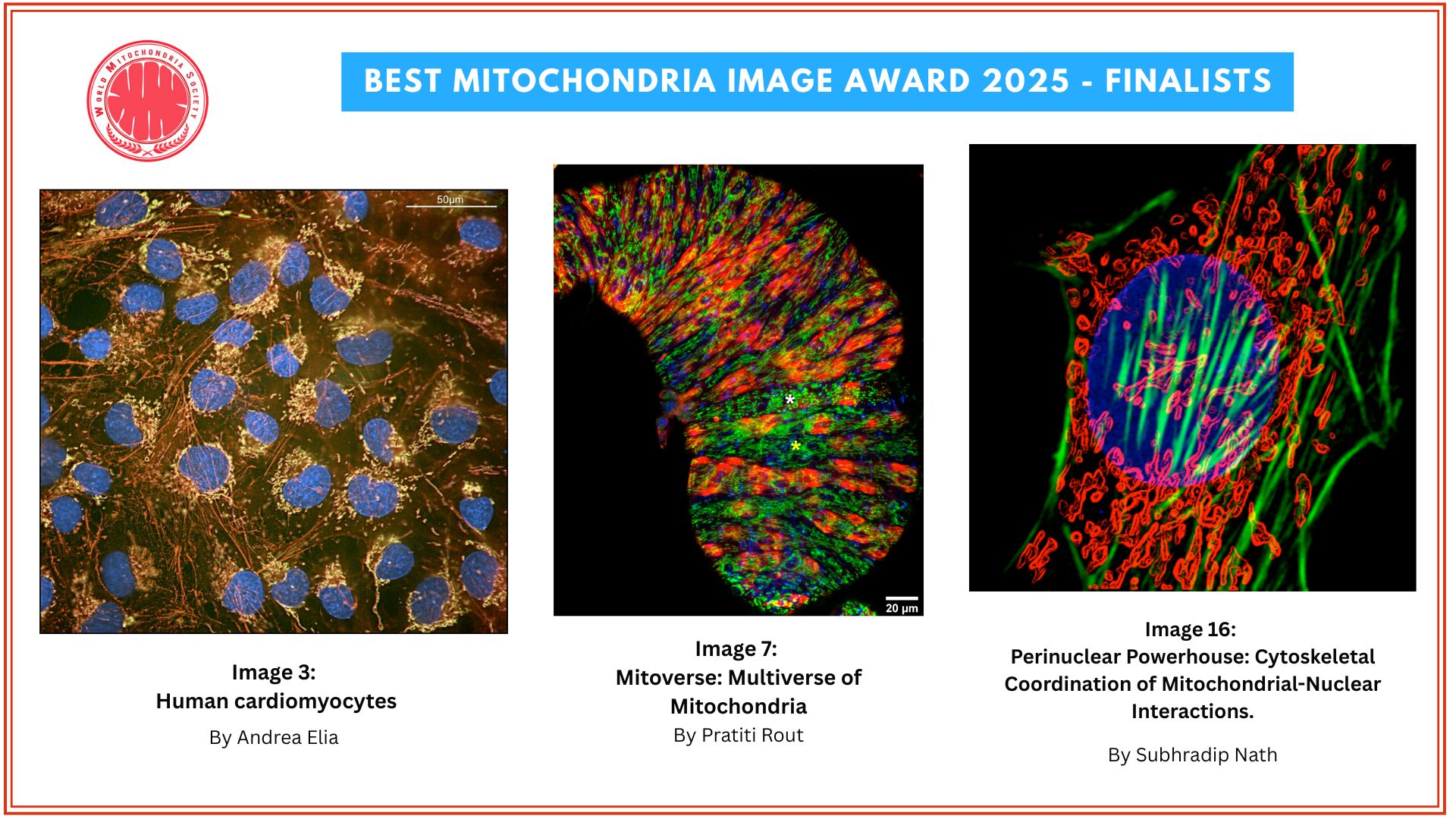
Best Image Award 2025 Winners Announced
- Details
- Published on 27 May 2025
We are thrilled to reveal the three winners of the Best Image in Mitochondria Research 2025!
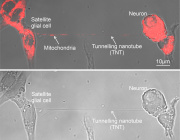
Mitochondria & Organelle Crosstalk - Rethinking Organelle Crosstalk: Mitochondrial-Derived Vesicles in Peroxisome Biogenesis Presented by Dr. Ayumu Sugiura
- Details
- Published on 19 May 2025

At the heart of cellular metabolism, mitochondria and peroxisomes play tightly interconnected roles in lipid regulation, redox homeostasis, and energy dynamics. While direct contacts between these organelles have long been observed, the mechanisms underlying their communication and biological significance are only beginning to emerge.
In an insightful presentation, Dr. Ayumu Sugiura of Juntendo University, Japan, introduces a compelling hypothesis: mitochondrial-derived vesicles (MDVs) may serve as essential mediators in peroxisome biogenesis. These vesicles, generated by mitochondria in response to cellular cues, could carry lipids, enzymes, or signaling molecules critical for initiating or modulating peroxisomal function.
“Mitochondrial-derived vesicles may provide a missing mechanistic link in understanding how mitochondria influence peroxisome formation and specialization,” says Dr. Sugiura.
His talk emphasized that this vesicular communication is not a byproduct of stress or degradation but a targeted and regulated form of inter-organelle signaling, reflecting a deeper evolutionary connection.
Understanding MDVs and their role in peroxisome biology may open new avenues in treating metabolic disorders, neurodegenerative diseases, and inherited mitochondrial syndromes, where organelle cooperation is often impaired.
This new perspective encourages scientists to rethink organelle crosstalk not as static interactions but as dynamic exchanges of molecular information, and places MDVs at the center of this emerging dialogue.
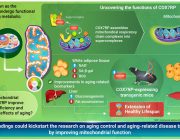
Targeting Mitochondrial Pyruvate Carrier: Impact on Future Metabolic Therapies
- Details
- Published on 22 April 2025
 Prof. Edmund Kunji from the University of Cambridge will give a major talk entitled Targeting mitochondrial pyruvate carrier: impact on future metabolic therapies, during the Targeting Mitochondria 2025 Congress, which will be held on October 22-24, in Berlin, Germany.
Prof. Edmund Kunji from the University of Cambridge will give a major talk entitled Targeting mitochondrial pyruvate carrier: impact on future metabolic therapies, during the Targeting Mitochondria 2025 Congress, which will be held on October 22-24, in Berlin, Germany.
About Prof. Kunji's talk:
Fifty years after the mitochondrial pyruvate carrier (MPC) was first identified, researchers have now resolved its molecular structure and mechanism of action. In a landmark study published in Science Advances, Sichrovsky et al. (2025) unveiled how this critical mitochondrial complex mediates pyruvate transport and how its inhibition could be leveraged for therapeutic purposes in cancer, metabolic disorders, and more.
About his outstanding study:
Major Discoveries of the Study by Prof. Edmund Kunji and his teams
Molecular Structure of MPC:
The authors used cryo-electron microscopy to capture the architecture of the human MPC complex. They discovered that MPC forms a heterodimeric transport unit (MPC1/MPC2), creating a selective channel that guides pyruvate across the inner mitochondrial membrane.
Mechanism of Transport and Inhibition:
The study revealed how small-molecule inhibitors bind to the MPC complex and block its function, offering a blueprint for drug development. Structural analysis pinpointed specific binding sites that explain both transport dynamics and inhibition sensitivity.
Conserved Functionality:
Evolutionary conservation of the MPC mechanism across species (including yeast and human) underscores its universal biological role in cellular energy homeostasis.
Therapeutic Implications
Cancer:
Some tumors overexpress MPC to fuel high mitochondrial activity. MPC inhibitors could starve these cells of essential metabolites, selectively disrupting their growth.
Metabolic Diseases:
In conditions like non-alcoholic fatty liver disease (NAFLD), blocking MPC forces hepatocytes to burn fat instead of relying on glucose, leading to reduced liver fat accumulation.
Regenerative Medicine & Hair Growth:
MPC inhibition has been shown to stimulate lactate production, which may promote hair follicle cell activation, opening potential new treatments for alopecia.
Mitochondrial Dysfunction & Neurodegeneration:
Targeting MPC may allow modulation of energy metabolism in neurodegenerative and mitochondrial diseases, where ATP production and redox balance are impaired.
Broader Impact
Drug Development:
The structural elucidation of MPC provides a molecular framework for designing selective modulators, setting the stage for new classes of metabolic drugs.
Precision Medicine:
Understanding individual differences in MPC structure/function may lead to personalized metabolic therapies tailored to genetic or disease-specific metabolic profiles.
Synthetic Biology & Bioenergetics:
The detailed MPC model can inform the engineering of customized metabolic pathways, supporting advances in synthetic biology, cell therapies, and biotechnology.
Keynote Speech: Targeting Mitochondrial Channels: Update and Strategies
- Details
- Published on 11 April 2025
 Keynote Speech: Targeting Mitochondrial Channels: Update and Strategies
Keynote Speech: Targeting Mitochondrial Channels: Update and Strategies
In his keynote speech, Prof. Szewczyk will provide the latest insights into the role of mitochondrial channels in cellular function and disease. He will discuss recent advancements and strategic approaches for targeting these channels, highlighting their potential in therapeutic interventions.
About Adam Szewczyk
Adam Szewczyk is a Professor of Biochemistry and former director of the Nencki Institute of Experimental Biology (Polish Academy of Sciences) in Poland, and since 2022, he has served as the President of the Polish Biochemical Society. He completed his chemical studies at Warsaw University and his postdoctoral fellowship at Bern University (Switzerland), Institute of Cellular and Molecular Pharmacology-Nice University (France), and at Johns Hopkins University, Baltimore, MD. He is the Head of the Laboratory of Intracellular Ion Channels at Nencki Institute.
His research is focused on the role of ion channels on mitochondrial function and intracellular ion channels pharmacology and biophysical properties of mitochondrial potassium channels.
World Mitochondria Society
Annual World Congress on Targeting Mitochondria
October 22-24, 2025 - Berlin, Germany
LinkedIn | Facebook
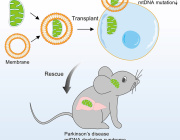
Development of Mitochondria-Based Therapeutic Strategies for Disease Treatment
- Details
- Published on 11 April 2025
 We are pleased to announce that Prof. Kosuke Kusamori from Tokyo University of Science, Japan, will be presenting his pioneering research on "Development of Mitochondria-Based Therapeutic Strategies for Disease Treatment."
We are pleased to announce that Prof. Kosuke Kusamori from Tokyo University of Science, Japan, will be presenting his pioneering research on "Development of Mitochondria-Based Therapeutic Strategies for Disease Treatment."
Summary
In recent years, the application of mitochondria isolated from cells for disease treatment has gained increasing attention, with their efficacy demonstrated in several diseases. However, the functions and characteristics of isolated mitochondria remain largely unknown, and their kinetics after administration into the body have yet to be fully elucidated.
Prof. Kusamori has been investigating the physical properties and in vivo kinetics of isolated mitochondria. In this talk, Prof. Kusamori will present his research on mitochondria-based therapeutic strategies aimed at advancing mitochondrial therapeutics.
World Mitochondria Society
Annual World Congress on Targeting Mitochondria
LinkedIn | Facebook
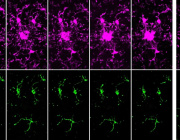
How Mitochondria Organize Their Powerhouse Machinery for Optimal Performance
- Details
- Published on 11 April 2025
 We are pleased to announce that Dr. Florent Waltz from the University of Basel, Switzerland, will be presenting at the Targeting Mitochondria 2025 congress in Berlin, Germany, on October 22-24, 2025.
We are pleased to announce that Dr. Florent Waltz from the University of Basel, Switzerland, will be presenting at the Targeting Mitochondria 2025 congress in Berlin, Germany, on October 22-24, 2025.
Dr. Waltz will share insights from his groundbreaking research on "How Mitochondria Organize Their Powerhouse Machinery for Optimal Performance" with a special focus on photosynthetic organisms.
Key Highlights:
- In Situ Visualization: Researchers employed advanced imaging techniques to observe the mitochondrial respiratory chain within intact cells, providing a detailed view of its native architecture.
- Respiratory Supercomplexes: The study offers insights into how respiratory complexes assemble into supercomplexes, which are crucial for efficient electron transport and energy production in cells.
- Functional Implications: Understanding the organization of these supercomplexes sheds light on their role in cellular metabolism and energy conversion, potentially informing research into mitochondrial-related diseases.
Perspective:
- Challenging previous assumptions: The findings challenge long-standing models that assumed a more fluid, random distribution of respiratory chain components in mitochondrial membranes.
- Biological relevance: By analyzing structures in situ, this study underscores the importance of studying macromolecular organization in native cellular contexts, rather than relying only on purified proteins.
- Broader implications: These insights are critical not only for basic mitochondrial biology but also for understanding mitochondrial dysfunction in aging, neurodegenerative diseases, and metabolic disorders.
- New model for mitochondrial function: This study supports a model in which the geometrical and biochemical compartmentalization within cristae contributes significantly to the efficiency of oxidative phosphorylation.
These findings enhance our comprehension of mitochondrial function and may have implications for addressing metabolic disorders linked to mitochondrial dysfunction.
About the Speaker:
Dr. Florent Waltz leads research at the University of Basel focusing on mitochondrial biology and evolution in photosynthetic organisms, particularly micro-algae. His laboratory employs state-of-the-art imaging technologies to reveal the intricate details of how these essential organelles function and adapt.
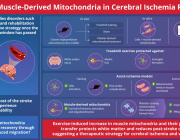
Prof. Yosuke Togashi to Present Groundbreaking Insights on Mitochondrial Transfer and Immune Evasion in Cancer
- Details
- Published on 10 April 2025

Prof. Togashi’s upcoming lecture follows his landmark publication in Nature (February 2025), where his team uncovered a novel immune evasion mechanism in cancer: the direct transfer of mutated mitochondria from cancer cells to tumor-infiltrating T cells (TILs).
Why Prof. Togashi’s Contribution Is Strategic
- Discovery of Mitochondrial Transfer as an Immune Evasion Mechanism
Prof. Togashi and colleagues demonstrated that cancer cells transfer mutated mitochondria to tumor-infiltrating T cells (TILs), leading to functional impairment of the immune response. - Homoplasmic Replacement in T Cells
This transfer causes replacement of healthy T cell mitochondria with cancer-derived, mutation-bearing mitochondria—effectively sabotaging T cell metabolism, memory formation, and anti-tumor function. - Inhibition of Mitophagy via USP30
He uncovered that mitophagy-inhibitory molecules, especially USP30, are transferred along with the mitochondria, preventing the degradation of dysfunctional mitochondria in T cells. - Impact on Immunotherapy Resistance
The study linked mitochondrial transfer to poor response to immune checkpoint inhibitors (ICIs) in melanoma and lung cancer patients, providing a novel biomarker and therapeutic target to overcome resistance. - Therapeutic Reversibility
Blocking mitochondrial transfer or inhibiting USP30 restored T cell function, paving the way for new combinatory approaches to boost immunotherapy efficacy.
A New Era in Cancer Immunometabolism
Prof. Togashi’s insights underscore the critical need to rethink cancer metabolism and immune resistance at the mitochondrial level. His work bridges cancer biology, immunology, and mitochondrial medicine—a core mission of the Targeting Mitochondria community.
More Articles...
- Fighting Fire with Fire: Boosting T Cell Therapy by Intercellular Mitochondrial Transfer
- Mitochondria and Creatine: Perspectives and Strategies
- Brain Organoids for drug discovery of Mitochondrial Neurological Disorders
- New Workshop on Mitochondrial Transfer and Transplantation: From Bench to Bedside